Modeling the Bonds of Iron and Water
February 28, 2011
Contact: Linda Vu, lvu@lbl.gov , 510.495.2402

Water lone pair orbitals in the first (left) and second (right) hydration shells of a high spin iron and aluminum. Image: © 2010 American Chemical Society.
Using supercomputers at the Department of Energy's National Energy Research Scientific Computing Center (NERSC), scientists have successfully modeled the atomic interactions between a high spin ferric iron ion and 64 water molecules for the first time.The model is based entirely on the fundamental equation of quantum mechanics, known as the Schrödinger equation; solving this equation is the only way to gain reliable insights into the behavior of atoms.
Researchers say this unprecedentedly accurate model will offer valuable insights into the process of underground mineral formation and extraction, as well as the transport of toxic materials in groundwater. By understanding these key reactions, scientists hope to develop effective strategies for carbon sequestration and other environmental remediation projects.
A paper on this work was published in the January 2010 Journal of Physical Chemistry A. The study was carried out by the group of John Weare (Chemistry and Biochemistry Department, University of California San Diego) in collaboration with researchers at Pacific Northwest National Laboratory. The project was led by Stuart Bogatko as part of his doctoral research. The team is currently validating these computer models with experiments at DOE's Advanced Photon Light Source facility and the Stanford Linear Accelerator Center; a paper on this work is forthcoming.
Powerful Computers Increase Scientific Accuracy
"We can use quantum mechanic equations to gain valuable insights about Earth’s chemistry by accurately modeling how metal ions and other materials react in water at different temperatures and pressures. But until this study, accurate on the fly representations of these reactions were difficult to obtain," says Eric Bylaska, of the Pacific Northwest National Laboratory and co-author on the Journal of Physical Chemistry paper.
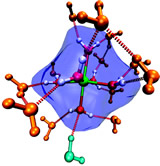
H-bond interaction between second shell waters and the first shell. The orange waters are trigonally H-bonded to the first shell (which is partially obscured to show the interactions more clearly). The light blue water is coordinated by a first shell acceptor bond.
Researchers have long known that the properties of a molecular system could be accurately predicted by solving the Schrödinger equation for all consisting atoms, but many choose to rely on approximate methods because they require less computing power. While this strategy works in many cases, Bylaska notes that scientific accuracy breaks down when surrounding variables like temperature or pressure change. Thus, it is not ideal for geochemistry research.
"If we ran our system of 64 water molecules and an iron ion on a desktop computer, it would take a couple of hours to solve one Schrödinger equation. But using supercomputers like NERSC's Franklin or Bassi, we can solve that same problem in less than one second," says Bylaska. "What we accomplished in a few hours on NERSC systems would have taken about 11 years to do on a desktop computer."
Using approximately 272 processors on NERSC’s Cray XT4 "Franklin" system, and about 112 processors on the facility's IBM POWER 5 "Bassi" system, Bogatko generated a simulation that lasted five times longer than any previous model. The system also contained twice as many water molecules as preceding studies.
Bylaska likens these simulations to making a movie. "Each Schrödinger equation is equivalent to one frame of a film. We have to solve these equations in less than a second to get enough snapshots to see what is going on, and this would not be possible without supercomputers," he adds. "As technology becomes increasingly sophisticated, we will be able to accurately model even larger systems in better detail."
In addition to Bogatko and Bylaska, John Weare of the University of California, San Diego was also an author on the paper. The NERSC allocation was granted by DOE's Office of Basic Energy Sciences. Bassi was decommissioned in April 2010 and replaced with an IBM iDataPlex system called "Carver."
About Computing Sciences at Berkeley Lab
High performance computing plays a critical role in scientific discovery. Researchers increasingly rely on advances in computer science, mathematics, computational science, data science, and large-scale computing and networking to increase our understanding of ourselves, our planet, and our universe. Berkeley Lab’s Computing Sciences Area researches, develops, and deploys new foundations, tools, and technologies to meet these needs and to advance research across a broad range of scientific disciplines.







 Instagram
Instagram YouTube
YouTube