Water and Gold: A Promising Mix for Future Batteries
Berkeley Lab Study Reveals Molecular Structure of Water at Gold Electrodes
October 23, 2014
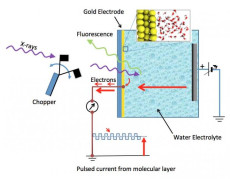
Schematic of the electrochemical cell – a silicon nitride (Si3N4) membrane separates the liquid from vacuum region of the x-ray source; a 20nm thin-film gold electrode is deposited on liquid side of the membrane. Detection of x-ray absorption is via fluorescence emission on the vacuum side or electron emission at the gold electrode. Image: Berkeley Lab
When a solid material is immersed in a liquid, the liquid immediately next to its surface differs from that of the bulk liquid at the molecular level. This interfacial layer is critical to our understanding of a diverse set of phenomena from biology to materials science. When the solid surface is charged, just like an electrode in a working battery, it can drive further changes in the interfacial liquid. However, elucidating the molecular structure at the solid-liquid interface under these conditions has proven difficult.
Now, for the first time, researchers at the US Department of Energy’s (DOE) Lawrence Berkeley National Laboratory (Berkeley Lab) have observed the molecular structure of liquid water at a gold surface under different charging conditions. Their findings were published October 23, 2014 in Science.
Lead author Miquel Salmeron, a senior scientist in Berkeley Lab’s Materials Sciences Division (MSD) and professor in UC Berkeley’s Materials Science and Engineering Department, and his team have developed a method not only to look at the molecules next to the electrode surface but to determine their arrangement changes depending on the voltage. Calculations run at the National Energy Research Scientific Computing Center (NERSC) by co-author David Pendergast, a staff scientist in the Lab's Molecular Foundry and researcher in the Joint Center for Energy Storage Research (JCESR), helped them better understand the chemistry involved.
“This is the first time that an experiment has been able to give this much detail about the molecular structure of a liquid near an interface—and a real liquid at that, not a small cluster of water molecules at cold temperatures, but liquid water at room temperature,” said Pendergrast. "This paves the way for future studies of electrochemical interfaces that exist in current and prototype batteries."
Spewing Photons and Electrons
With gold as a chemically inert electrode, and slightly-saline water as an electrolyte, Salmeron and colleagues used a new twist on x-ray absorption spectroscopy (XAS) to probe the interface and show how the interfacial molecules are arranged. XAS itself is not new. In this process, a material absorbs x-ray photons at a specific rate as a function of photon energy. A plot of the absorption intensity as a function of energy is referred to as a spectrum which, like a fingerprint, is characteristic of a given material molecule and its chemical state. Our eyes recognize many materials by their characteristic colors, which are related to their visible light absorption spectra. The x-ray photons used in this study have energies that are about 250 times higher than those of visible light and are generated at Berkeley Lab’s Advanced Light Source (ALS).
Typical XAS measurements are made under vacuum conditions, as x-rays are readily absorbed by matter, even the nitrogen molecules in air. But liquids will quickly evaporate in a vacuum. By using a very thin (100 nm, or a tenth of a micrometer) x-ray transparent window, with a thin coating of gold (20nm), on a sealed liquid sample holder, the Berkeley Lab team was able to expose water molecules in the liquid to x-rays and collect their spectra.
Upon absorbing an x-ray photon, the excited water molecule can spew (emit) either charged particles (electrons) or light (photons). The amount of photon emission, or fluorescence, is one indicator of how many x-ray photons have been absorbed. However, fluorescing x-rays can be detected from molecules ranging from those at the gold surface to those deep (micrometers) inside the liquid far from the influence of the gold surface, and these dominate the measured spectrum.
“We are only really interested in a nanoscale interfacial region, and looking at the fluorescence photon signal we can’t tell the difference between the interface and the interior electrolyte molecules,” said Salmeron.
Measuring Electron Emissions
The challenge therefore was to collect a signal that would be dominated by the interfacial region. The team accomplished this by measuring electron emissions because electrons emitted from x-ray excited water molecules travel only nanometer distances through matter. The electrons arriving at the gold electrode surface can be detected as an electrical current traveling through a wire attached to it. This avoids confusion with signals from the interior electrolyte because electrons emitted from interior molecules don’t travel far enough to be detected.
There’s an additional problem that arises when studying liquids in contact with working electrodes because they carry a steady current as in batteries and other electrochemical systems. While the emitted electrons from nearby molecules are indeed detectable, this contribution to the current is dwarfed by the normal “Faradaic” current of the battery at finite voltages. When measuring current off the electrode, it is critical to determine which part is due to the x-rays and which is due to the regular battery current.
To overcome this problem, the researchers pulsed the incoming x-rays from the synchrotron at a known frequency. The current contribution resulting from electron emission by interfacial molecules is thus pulsed as well, and instruments can separate this nanoampere modulated current from the main Faradaic current.

Left to right: Miquel Salmeron, Jinghua Guo, David Prendergast, Liwen Wan, Chenghao Wu, Tod Pascal and Juan-Jesus Velasco-Velez. Their ground-breaking study, “The structure of interfacial water on gold electrodes studied by x-ray absorption spectroscopy,” was published October 23, 2014, in Science. Photo: Roy Kaltschmidt, LBNL
These experiments result in absorption vs. x-ray energy curves (spectra) that reflect how water molecules within nanometers of the gold surface absorb the x-rays. To translate that information into molecular structure, a sophisticated theoretical analysis technique is needed.
From Spectra to Structures
Prendergast developed the computational techniques that enabled this translation. Using the Hopper supercomputer at NERSC, he conducted large molecular dynamics simulations of the gold-water interface and then predicted the x-ray absorption spectra of representative structures from those simulations.
“These are first-principles calculations,” explained Prendergast. “We don't dictate the chemistry: we just choose what atomic elements are present and how many atoms. That’s it. The chemistry is a result of the calculation.”
It turns out that for a neutral gold surface, a significant number of water molecules (H2O) next to the gold surface orient with hydrogen (H) atoms pointing toward the gold. Water molecules are bound together by so-called hydrogen bonds, which orient the slightly positively-charged H atoms in each molecule towards the slightly negatively-charged oxygen (O) atoms of neighboring molecules. This network of hydrogen bonds is what holds water molecules together to make a liquid under conditions of temperature and pressure that we consider comfortable as humans. It is perhaps surprising that the inert gold surface can induce significant numbers of water molecules not to hydrogen-bond to each other but to bond to the gold instead. This number is enhanced when the gold is negatively charged and therefore attracting the more positive H atoms. Furthermore, positively-charged gold ions cause water molecules to orient their H atoms away from the gold, which strengthens the hydrogen bond network of the interfacial liquid.
“That’s the main thing we know about the gold electrode surface from the x-ray absorption spectra: how many water molecules are tilted one way or another, and if their hydrogen bonds are broken or not,” concluded Salmeron. “Water next to the electrode has a different molecular structure than it would in the absence of the electrode.”
There are a couple of subtle things that are very important, added Prendergast. First, the shape of the absorption spectra changes as a function of changing voltage. Since the measured spectra agree with the calculations one can draw conclusions about the molecular structure of the liquid interface as a function of voltage. The second is that in the calculations, the change in the structure of water is limited to the first two molecular layers above the surface and these two layers span only about 1 nanometer. To observe any difference in the experimental spectra with varying voltage means that measurements are sensitive to a shorter length scale than was thought possible.
“We had thought the sensitivity to be tens of nanometers, but it turns out to be subnanometer,” said Prendergast. “That’s spectacular!”
This research was primarily supported by the DOE Office of Science. The ALS, the Molecular Foundry and NERSC are all DOE Office of Science User Facilities. JCESR is an Energy Innovation Hub funded by DOE’s Office of Science.
About Computing Sciences at Berkeley Lab
High performance computing plays a critical role in scientific discovery. Researchers increasingly rely on advances in computer science, mathematics, computational science, data science, and large-scale computing and networking to increase our understanding of ourselves, our planet, and our universe. Berkeley Lab’s Computing Sciences Area researches, develops, and deploys new foundations, tools, and technologies to meet these needs and to advance research across a broad range of scientific disciplines.







 Instagram
Instagram YouTube
YouTube